The mission of the West Laboratory is the development of novel therapeutics for the treatment of traumatic brain injury (TBI) and stroke. TBI and stroke are leading causes of long-term disability and death in the United States. The West Laboratory focuses on regenerative therapies that promote neurogenesis, angiogenesis and activation of neuroprotective pathways with an emphasis on stem cell therapies. The West Laboratory breakthrough discoveries have shown that patient derived neural stem cells can replace lost neural cells and regenerate and stabilize damaged brain tissue leading to improvements in motor function, behavior, and cognition. The West Laboratory also develops magnetic resonance imaging (MRI) brain structural and functional assessment technologies to better predict learning, memory, and motor function deficits and long-term prognosis for patients that have suffered from TBI and stroke.
Follow us on Instagram
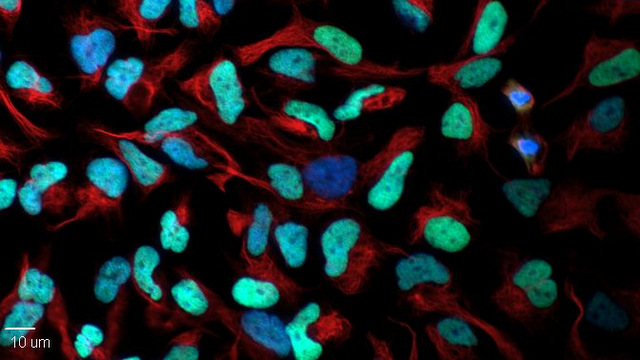
Bergann’s TBI therapeutic results showed significant improvement in diffusivity and white matter integrity. Truly some amazing work that she’s done this year!

Regenerative Biosciences powered by Taco Bell! We got the Tacos, the Gorditas, and the Chalupa! 🧪🌮🧫🌮🔬

Drew showing some of the faculty all her hard work from this year! Can’t wait to see what you do in graduate school!

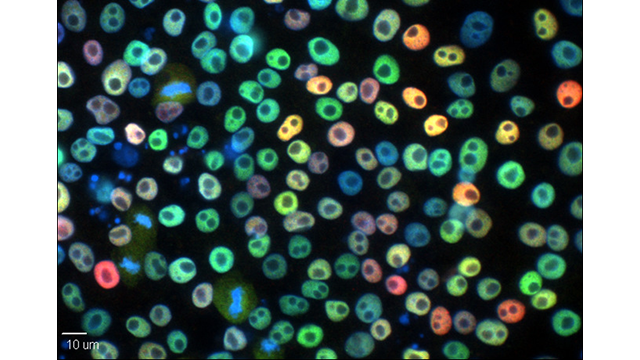
Sarah shared some phenomenal results pertaining to a novel traumatic brain injury therapeutic at the RBC ADS Research Symposium! 🧫🧑🔬

It’s been a few years since I took the original picture! But the love for what we do hasn’t changed!

I went to check my mailbox and I found this guy in there! We used to only have pigs coming out of our ears. Now they’re coming out of our mailbox!

Come learn more about cutting edge biomedical imaging in pig models to help millions of patients with neurological conditions. We are going to be there. How about you? https://sbr2024.org #TNRRlab #westlab

Congratulations to one of our amazing undergraduate researchers, Kristy Trung, for getting into the UGA Vet School! We’re so proud of you and you’re amazing accomplishment!

A huge congratulations to Moira for being selected as a Franklin Foundation Scholar! What an accomplishment and a transformative research opportunity! Thank you to the Franklin Foundation and UGA BIRC.

Congratulations to Sarah Schantz for being selected for the John and Mary Franklin Foundation travel award!. San Francisco, Neurotrauma 2024 conference here we come!